 | |
| Clinical data | |
|---|---|
| Trade names | Aphexda |
| Other names | BL-8040 |
| License data | |
| Routes of administration | Subcutaneous |
| Drug class | Antineoplastic |
| ATC code |
|
| Legal status | |
| Legal status | |
| Identifiers | |
| CAS Number |
|
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII |
|
| KEGG | |
| ChEBI | |
| Chemical and physical data | |
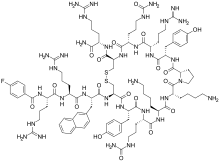
| Formula | C97H144FN33O19S2 |
| Molar mass | 2159.55 g·mol−1 |
Motixafortide, sold under the brand name Aphexda, is a medication used for the treatment of multiple myeloma.[1] Motixafortide is a hematopoietic stem cell mobilizer and a CXCR4 antagonist.[1] It is given by subcutaneous injection.[1]
Motixafortide was approved for medical use in the United States in September 2023.[3][4]
Medical uses
Motixafortide is indicated in combination with filgrastim, a granulocyte-colony stimulating factor (G-CSF), to mobilize hematopoietic stem cells to the peripheral blood for collection and subsequent autologous transplantation in people with multiple myeloma.[1][3]
Society and culture
Names
Motixafortide is the international nonproprietary name.[5]
References
- 1 2 3 4 5 "Aphexda- motixafortide injection, powder, lyophilized, for solution". DailyMed. 4 September 2023. Archived from the original on 14 September 2023. Retrieved 13 September 2023.
- ↑ "Aphexda- motixafortide acetate injection, powder, lyophilized, for solution". DailyMed. 22 September 2023. Retrieved 6 October 2023.
- 1 2 "Aphexda approval letter" (PDF). 8 September 2023. Archived from the original (PDF) on 14 September 2023.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ↑ "BioLineRx Announces FDA Approval of Aphexda (motixafortide) in Combination with Filgrastim (G-CSF) to Mobilize Hematopoietic Stem Cells for Collection and Subsequent Autologous Transplantation in Patients with Multiple Myeloma" (Press release). BioLineRx Ltd. 11 September 2023. Retrieved 13 September 2023 – via PR Newswire.
- ↑ World Health Organization (2019). "International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 82". WHO Drug Information. 33 (3). hdl:10665/330879.
Further reading
- Crees ZD, Rettig MP, Jayasinghe RG, Stockerl-Goldstein K, Larson SM, Arpad I, et al. (April 2023). "Motixafortide and G-CSF to mobilize hematopoietic stem cells for autologous transplantation in multiple myeloma: a randomized phase 3 trial". Nature Medicine. 29 (4): 869–879. doi:10.1038/s41591-023-02273-z. PMC 10115633. PMID 37069359.
External links
- Clinical trial number NCT03246529 for "A Phase III, Safety, Tolerability and Efficacy of Combination Treatment of BL-8040 and G-GSF as Compared to Placebo and G-CSF for thE MobilizatioN of HematopoiEtic Stem Cells for Autologous TransplantatIon in SubjectS With MM (GENESIS)" at ClinicalTrials.gov
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.